The Good
👍 CMS added 11 additional surgical procedures to the ASC Covered Procedures List (CPL) in addition to the original 26 dental codes added in the proposed rule, including total shoulder arthroplasty and ankle reconstruction. Our comments can really make a difference! Also, the collection burden for the centers remains the same in 2024 as in 2023, with only a few minor changes. One was to update the age range for the colonoscopy measure to 45-70. The other was the definition of “up-to-date” vaccine status.
👍 CMS finalized its proposal to continue to align the ASC update factor with the one used to update HOPD payments, extending the five-year interim period an additional two calendar years (CY) through 2025. The extension of this policy results in an effective update of 3.1 percent for ASCs. This is an increase of 0.3 percent from the proposed rule. Please note that this is an average. Updates might vary significantly by code and specialty.
The Bad
👎 Despite requests from the ASC community to discontinue or limit the reporting of COVID-19 vaccine status, CMS has decided to continue the laborious requirement to track employees’, clinicians’, and other center non-employee’s vaccine status. They also proposed to update the numerator to specify the timeframes within which healthcare providers are considered up to date with CDC-recommended COVID-19 vaccines, including updated vaccine doses, beginning with the CY 2024 reporting period. The term “up-to-date” is defined as meeting the CDC’s criteria on the first day of the applicable reporting quarter. The current definition of “up-to-date” for the COVID-19 vaccination can be found at: https://www.cdc.gov/nhsn/pdfs/hps/covidvax/UpToDateGuidance-508.pdf.
The Ugly
🔄 Looming on the horizon: In 2025, new or new mandatory measures will require extensive and expensive data collection from in-house and outside vendors. OAS CAHPS will be mandatory in 2025. It is highly recommended that you begin your journey to find the right vendor and gather your statistics. Doing so will allow you to work on some potential negative data before it is loaded on the CMS website and train your staff to improve your scores. Also, in 2025, CMS proposes gathering data on THR and TKR. This data will be collected from the patients and will require cooperation with the provider’s office and perhaps an outside vendor.
🔄 Additionally, CMS modified several measures for the ASC Quality Reporting Program: CY 2024 reporting period and CY 2026 payment determination. Changes include:
• Updating the COVID-19 Vaccination Coverage Among Healthcare Personnel measure to utilize the term “up to date.”
• Modifying the survey instruments for the Cataracts Visual Function measure.
• Aligning the Colonoscopy Follow-Up Interval measure with current clinical guidelines.
🔄 CMS also finalized measures for the CY 2024 reporting period/CY 2026 payment determination, including Patient Burn, Patient Fall, Wrong Site, Wrong Side, Wrong Patient, Wrong Procedure, Wrong Implant, All-Cause Hospital Transfer/Admission, Cataracts Visual Function, Facility 7-Day Risk-Standardized Hospital Visit Rate after Outpatient Colonoscopy, Normothermia Outcome, Unplanned Anterior Vitrectomy, Hospital Visits after Orthopedic Ambulatory Surgical Center Procedures, Hospital Visits after Urology Ambulatory Surgical Center Procedures, and Facility-Level 7-Day Hospital Visits after General Surgery Procedures.
🔄 CMS adopted a new measure, the ASC-21: Risk-Standardized Patient Reported Outcome-Based Performance Measure (PRO-PM) Following Elective Primary Total Hip Arthroplasty (THA) and/or Total Knee Arthroplasty (TKA) in the ASC Setting (THA/TKA PRO-PM). The measure will have voluntary reporting periods in CY 2025, CY 2026, and CY 2027, followed by mandatory reporting beginning with the CY 2028 reporting period for the CY 2031 payment determination. Mandatory reporting would start on January 1, 2027.
🔄 CMS chose not to readopt ASC-7 Facility Volume Data on Selected ASC Surgical Procedures at this time but may choose to do so in the future.
What do you need to know about 2024 data collections?
Data Collected by the Center:
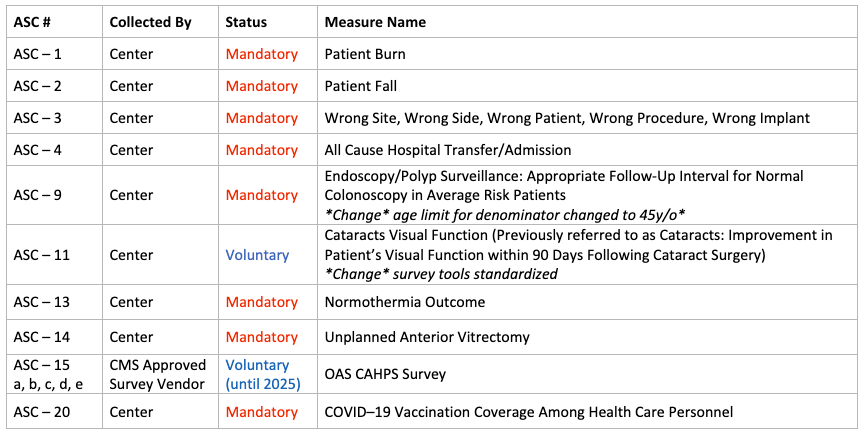
Data Collected by CMS:
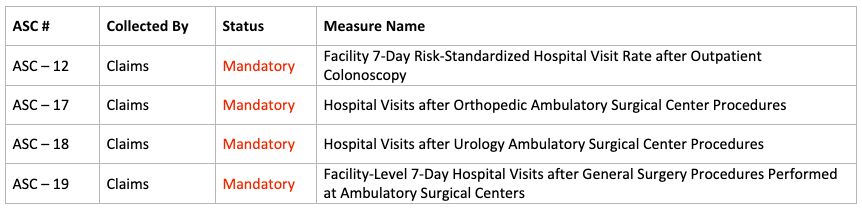
Vigilance and Adaptability are Key
As the ASC community navigates the complexities of the 2024 Final Payment Rule, it becomes evident that vigilance and adaptability are key. The expansion of ASC-covered procedures signifies progress, offering affordable options to patients and easing the financial burden on the Medicare program. However, the persistence of laborious reporting requirements and the looming mandatory measures in 2025 present challenges that demand proactive strategies. ASCs must focus on collaboration, research, and informed decision-making, embracing the opportunity to enhance patient care and streamline operations. By staying informed, engaging with regulatory changes, and leveraging collective voices, ASCs can continue shaping the healthcare landscape, ultimately ensuring a future marked by improved patient outcomes and sustainable healthcare practices.
Don’t miss out on the good stuff – Subscribe to HST’s Blog & Podcast!
Every month we’ll email you our newest podcast episodes and articles. No fluff – just helpful content delivered right to your inbox.
